Genaue und effiziente Mikroskopie-Bildanalyse mit TruAI und Deep Learning
Einführung
Untersuchungen erfordern häufig Daten von Mikroskopbildern. Zur genauen Bildanalyse ist eine Segmentierung wichtig, um den Zielbereich der Analyse aus dem Bild zu extrahieren. Eine gängige Segmentierungsmethode ist die Anwendung von Schwellenwerten für die Intensität oder die Farbe des Bildes.
Dies kann jedoch zeitaufwändig sein und den Probenzustand beeinträchtigen. Bildanalyseverfahren der nächsten Generation wie unsere Bildverarbeitungssoftware cellSens mit TruAI und Deep-Learning reduzieren die Risiken einer Probenschädigung und erreichen gleichzeitig eine hohe Effizienz und Genauigkeit.
Anwendungsbeispiele mit TruAI
1) Markerfreie Kerndetektion und Segmentierung
Um die Anzahl der Zellen zu zählen, Kerne in Zellen und Geweben zu lokalisieren und die Zellfläche zu bewerten, nutzen Forscher üblicherweise die Fluoreszenzfärbung von Kernen, um diese anhand der Fluoreszenzintensität zu segmentieren.
Im Gegensatz dazu kann TruAI die Zellkernsegmentierung mit Hellfeldbildern durchführen. Dazu wird mit den Ergebnissen der Kernsegmentierung aus Hellfeld- und Fluoreszenzbildern ein neuronales Netz trainiert.
Dieses selbstlernende Mikroskopiekonzept macht eine Fluoreszenzfärbung des Zellkerns überflüssig, sobald das neuronale Netzwerk erstellt ist. Weitere Vorteile:
- Minimierung des Zeitaufwands für die Zellkernfärbung
- Keine Beeinträchtigung der Zellen durch Anfärben
- Keine Fototoxizität, kein Ausbleichen
- Erfassung zusätzlicher Probeninformationen durch Ergänzung eines weiteren Kanals
 Label free nucleus detection by TruAI Abbildung 1 |
Abbildung 2 |
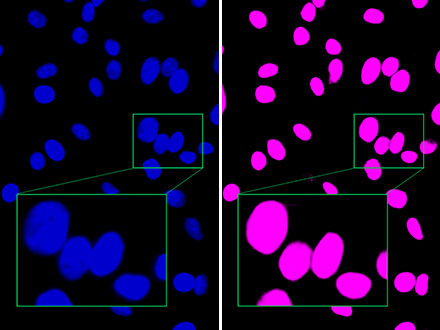
Abbildung 1: Während das Hellfeldbild (links) aufgrund der ungefärbten Zellen einen minimalen Kontrast aufweist, erkennt TruAI die Zellkerne mit hoher Genauigkeit (rechts).
Abbildung 2: Im Vergleich zum Fluoreszenzbild (links) unterscheidet TruAI von Olympus nahe beieinander liegende Zellkerne deutlich voneinander (rechts) und erlaubt eine Erkennung mit hoher Genauigkeit.
2) Quantitative Analyse von fluoreszenzmarkierten Zellen bei ultraschwachem Licht
Fluoreszierende Marker sind wertvolle Tools für moderne mikroskopische Zellstudien. Das dazu erforderliche intensive Anregungslicht kann jedoch zu Lichtschäden oder Fototoxizität führen und die Lebensfähigkeit der Zellen nachweislich beeinträchtigen. Auch wenn keine direkte Wirkung zu beobachten ist, kann starke Lichteinwirkung das natürliche Verhalten der Zellen beeinflussen und zu unerwünschten Effekten führen.
Bei Langzeitexperimenten mit lebenden Zellen ist eine minimale Lichtexposition während der Fluoreszenzmikroskopie ideal. Aus technischer Sicht erfordert die Belichtung mit extrem wenig Licht die Analyse von Bildern mit sehr niedrigem Signalpegel und folglich geringem Signal-Rausch-Abstand. Mit TruAI von Olympus lassen sich Bilder mit schwachen Signalen robust und präzise analysieren.
Abbildung 3 |
Abbildung 4 |
Abbildung 5 |
Abbildung 3: Das Ergebnis der Erkennung von Zellkernen (rechts) in einem Fluoreszenzbild (links) mit ausreichender Leuchtdichte mit einer herkömmlichen Methode (Auswertung einer Leuchtdichteschwelle).
Abbildung 4: Das Ergebnis der Erkennung von Zellkernen (rechts) mit der gleichen konventionellen Methode wie in Abbildung 3 in Fluoreszenzbildern (links) mit extrem niedrigem Signal-Rausch-Abstand aufgrund des schwachen Anregungslichts. Die geringe Erkennungsgenauigkeit ist offensichtlich.
Abbildung 5: Das Ergebnis der Erkennung von Zellkernen (rechts) mit TruAI in einem Fluoreszenzbild (links) mit extrem niedrigem Signal-Rausch-Abstand aufgrund des schwachen Anregungslichts. Es ist gut erkennbar, dass die Genauigkeit genauso hoch ist wie in Abbildung 3 und deutlich höher als in Abbildung 4.
3) Segmentierung nach morphologischen Merkmalen
Mit dem herkömmlichen Ansatz (Schwellenwerte für Intensität und Farbe) ist es sehr schwierig, eine hochpräzise Segmentierung eines Bilds auf der Grundlage morphologischer Merkmale zu erreichen. Daher mussten Zählung und Messung jedes Mal manuell durchgeführt werden.
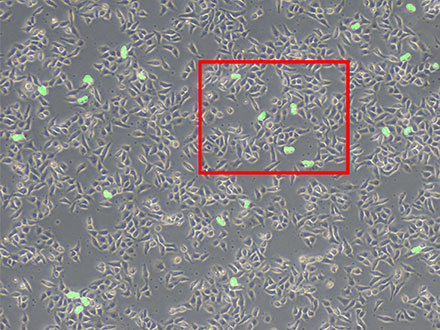
Im Gegensatz dazu ermöglicht TruAI eine hocheffiziente und genaue Segmentierung nach morphologischen Merkmalen. Sobald das neuronale Netz die Segmentierungsergebnisse aus manuell markierten Bildern gelernt hat, kann es die gleiche Methodik auf weitere Datensätze anwenden. Zum Beispiel können neuronale Netze, die mit manuell markierten Bildern trainiert wurden, mitotische Zellen zählen (siehe nachstehendes Bildmaterial).
Abbildung 6 |  A high-magnification image (left) of the framed area in Figure 6 Abbildung 7 |
Abbildung 6: Vorhersage mitotischer Zellen mit TruAI (grün).
Abbildung 7: Es werden zwar viele Zellen angezeigt, aber nur die sich teilenden Zellen werden erkannt (rechts).
4) Segmentierung von Gewebeproben
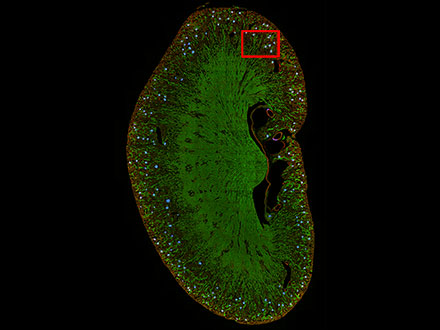
TruAI kann auch zur Segmentierung von Gewebeproben verwendet werden. Zum Beispiel können Nierenglomeruli, die mit herkömmlichen Methoden schwer zu unterscheiden sind, mit TruAI segmentiert werden.
Abbildung 8 |  A high-magnification image (left) of the framed area in Figure 8 Abbildung 9 |
Abbildung 8: Vorhersage von Glomeruli-Positionen auf einem Maus-Nierenschnitt mit TruAI (blau).
Abbildung 9: TruAI erfasst und detektiert die Glomeruli-Merkmale (rechts).
Zusammenfassung
Herkömmliche Segmentierungsmethoden können problematisch sein und die Proben schädigen. Unsere cellSens-Bildgebungssoftware mit Deep Learning ermöglicht eine genaue und effiziente Segmentierung unter zellschonenden Bedingungen, beispielweise durch markerfreie Bildgebung oder ultraschwache Beleuchtung. Die Software erleichtert auch die Segmentierung von Gewebeproben anhand ihrer morphologischen Merkmale.
Verwendete Produkte
wurde erfolgreich zu Ihren Lesezeichen hinzugefügt
Maximum Compare Limit of 5 Items
Please adjust your selection to be no more than 5 items to compare at once
Not Available in Your Country
Sorry, this page is not
available in your country.