In modern life science research, fluorescence microscopy is a popular and powerful imaging technique. With the fluorophores specifically labeled on a target, the emitted fluorescence signal can provide superb imaging contrast with minimal background down to the molecular level.
Highly developed optical technologies, as well as extensive labeling approaches from cutting-edge genetic engineering, are the success factors of fluorescence microscopy in biological research.
Yet, despite its overall success, the need for fluorescent labels or molecules still limits the applications of fluorescence microscopy:
- Many biomolecules are nonfluorescent, small, and easily perturbed by fluorescent labels
- The expression level and specificity of fluorophores in living organisms sometimes complicate the experimental results
- The use of exogenous fluorescent labels raises concerns on biomedical research in humans
Given these limitations, optical imaging methods using contrast other than fluorescence are in demand. In this post, we’ll share alternative biological imaging methods to help you think beyond fluorescence microscopy and expand your research capabilities.
Thinking Beyond Fluorescence Microscopy
Nonlinear optics can generate signals from intrinsic properties of specific molecules, forming imaging contrast without the need for fluorescence.
Thanks to the availability of advanced laser sources and microscopy techniques, nonlinear optics isn’t just for fancy experiments in the lab. It has become a feasible solution for biological imaging, inspiring researchers to think beyond fluorescence microscopy. One good example is optical harmonic generation (HG) microscopy.
Understanding SHG and THG Microscopy
Simply put, HG is a nonlinear optical process in which n photons simultaneously interact with the material and convert into one photon. Second harmonic generation (SHG, two photons into one photon) and third harmonic generation (THG, three photons into one photon) are the most commonly used HG microscopy in biological imaging.
The photon conversion process in SHG and THG is illustrated in the Jablonski diagram (Figure 1) below: SHG gives photons with double photon energy (half wavelength), and THG gives photons with triple photon energy (one-third wavelength) of excitation photons.
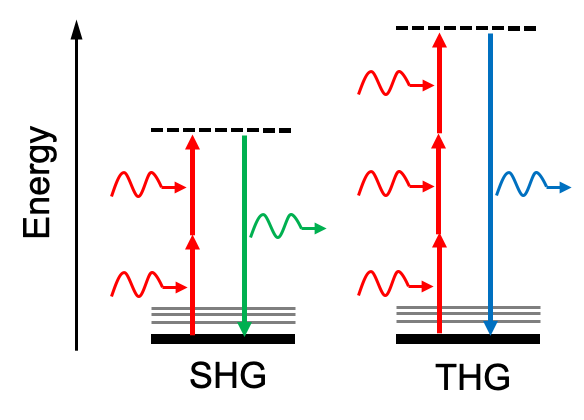
Figure 1: Jablonski diagram of SHG and THG optical processes. Dashed lines represent virtual states.
Like multiphoton microscopy, SHG and THG require an ultrafast pulsed laser source, typically in the near-IR wavelength range, to fulfill nonlinear optical process in a small excitation focal volume. As a result, SHG and THG microscopy have the following features:
- Intrinsic optical sectioning capability
- Deep imaging in highly scattering tissues
- Low photobleaching and phototoxicity
- SHG and THG signal intensities are proportional to the square and cubic of the laser excitation power, respectively
Yet, unlike multiphoton microscopy, SHG and THG signals come from the optical property and the structure of the material:
- SHG microscopy is commonly applied to the imaging of non-centrosymmetric molecules and ordered structures, such as collagen fibers, microtubules, muscle myosin, starch, skin tissues, and stroma of cornea.
- THG microscopy is usually applied to the imaging of high refractive index substances or interfaces (compared to surrounding water), such as cell organelles, red or white blood cells, lipid droplets, adipose tissue, axon myelin sheath, and bone.
Without the need for fluorophores, SHG and THG microscopy can achieve label-free imaging and provide molecular and structural information in biological tissues, living organisms, and even in medical imaging for diagnosis.
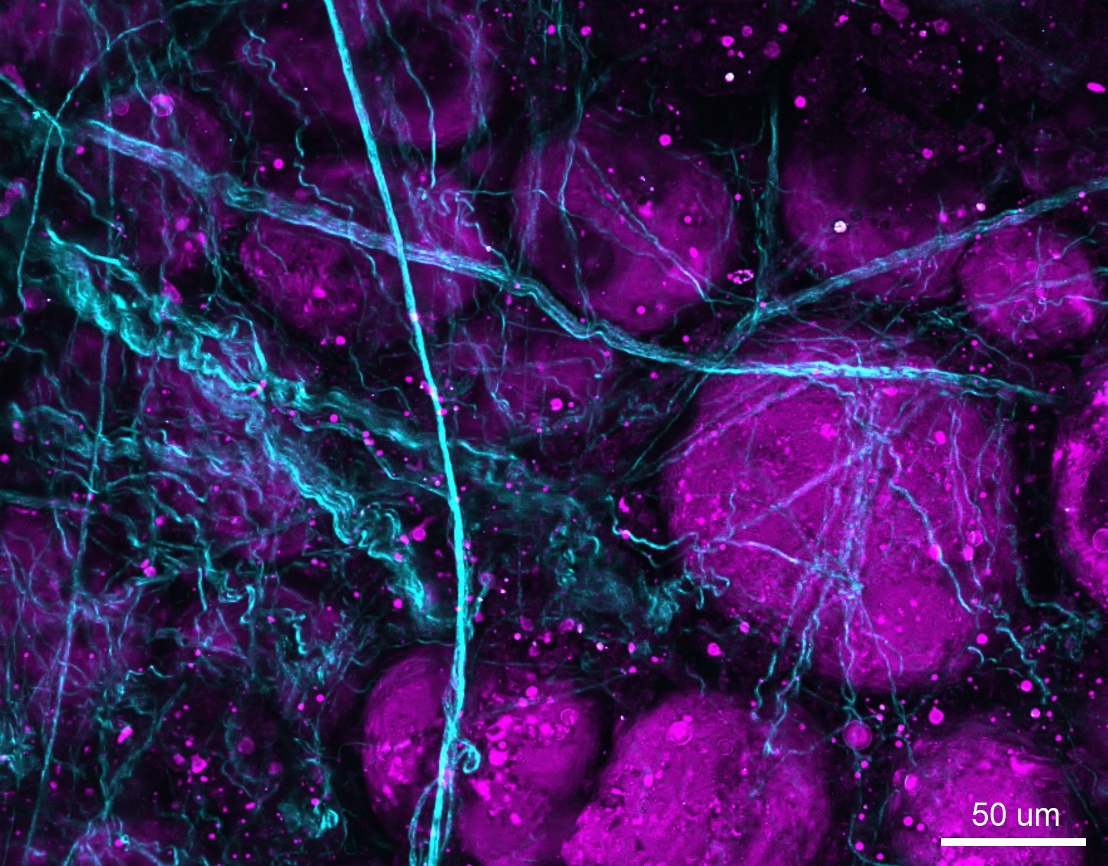
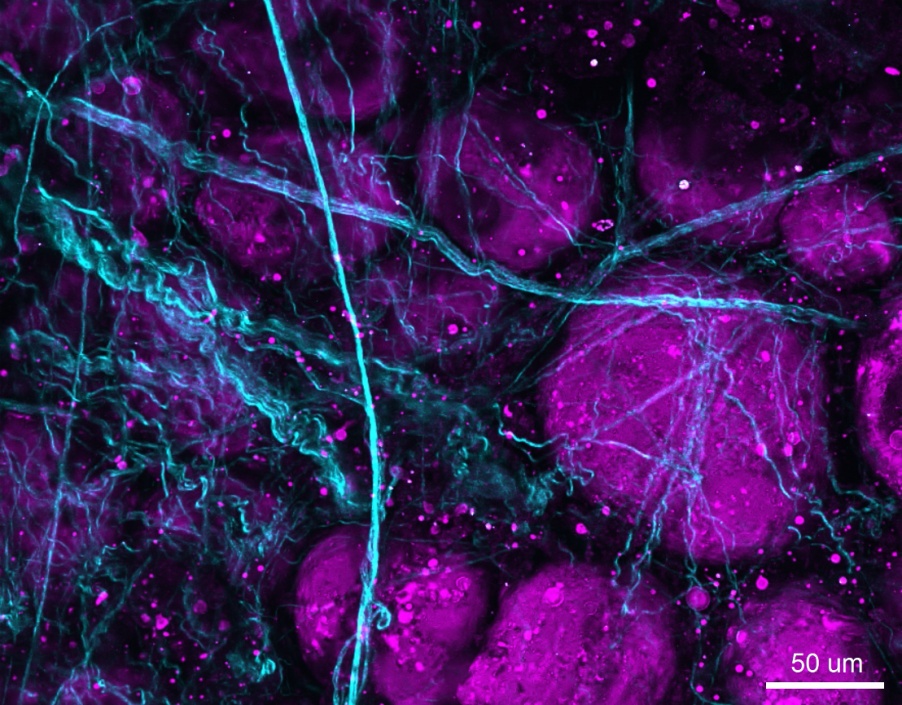
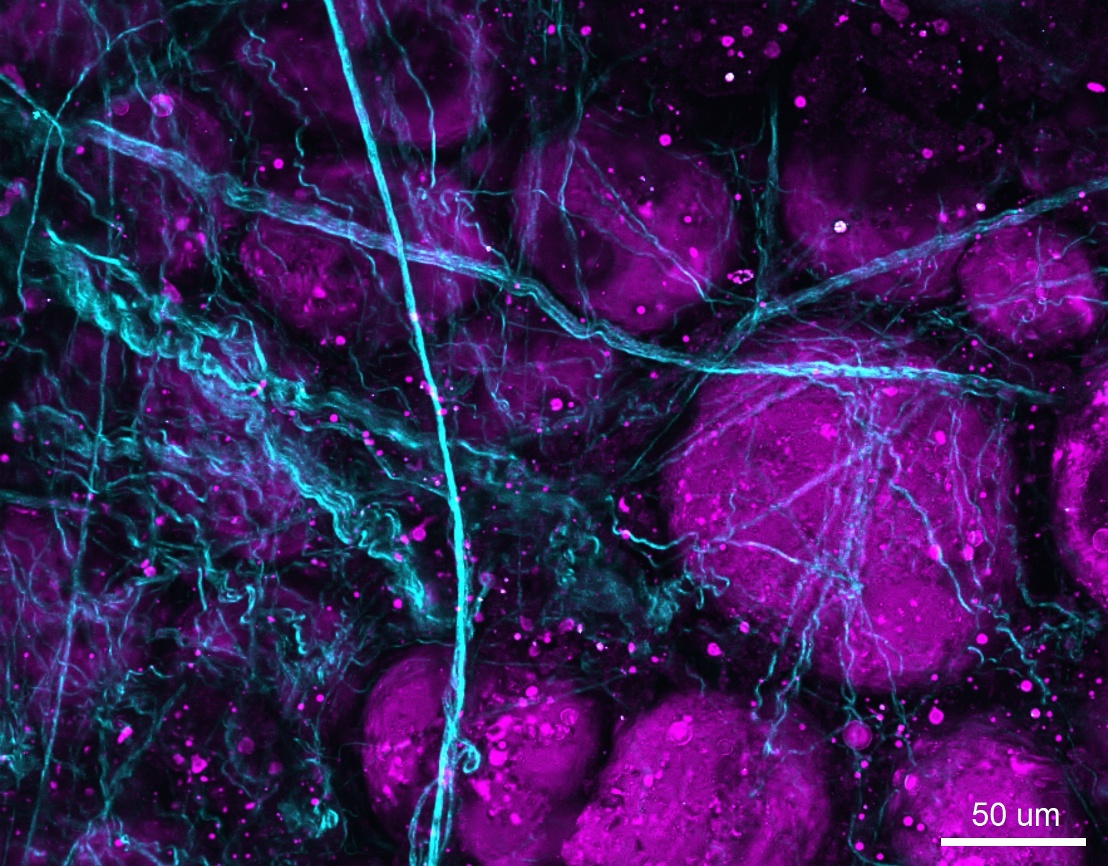
Figure 2: SHG/THG image of unlabeled porcine adipose tissue acquired using the Olympus FVMPE-RS multiphoton microscope. SHG signal (cyan) shows collagen fibers, and THG signal (magenta) shows fat in adipose tissue.
Now that we’ve covered the basics of SHG and THG microscopy, our next section will provide some tips to help you put these techniques into practice.
Practical Considerations for Configuring SHG and THG Microscopy
Because SHG and THG microscopy can share the same NIR-pulsed laser source and laser scanning unit with multiphoton microscopy, our FVMPE-RS multiphoton microscope readily combines with SHG and THG microscopy to create a multimodal imaging system.
Here are two practical considerations when setting up SHG and THG microscopy:
- Forward detection and high-sensitivity detectors:
While fluorescence emission is generally isotropic, SHG and THG signals have certain directionality originating from the wave-mixing process. In most samples, the forward signal (away from the objective) is much stronger than the backward signal (toward the objective). In this case, a high-NA condenser and transmitted non-descanned detectors (NDDs) are a good choice for forward signal detection. In highly scattering tissues, some forward signal can be backscattered and detected by backward signal detection. In this case, use high-sensitivity NDDs such as a gallium arsenide phosphide photomultiplier tube (GaAsP PMT) to optimize your backward detection efficiency, especially when forward signal detection is not applicable in bulk samples or live animals.
- Good laser power at a longer wavelength:
THG signal requires good laser excitation power to fulfill the nonlinear optical process and is generated at one third of the excitation wavelength. For better signal transmission and detection in THG microscopy, use a laser source with good power at a wavelength longer than 1200 nm so that the THG signal can fall within the visible light range. In the past, an NIR-pulsed laser, an optical parametric oscillator (OPO), and an expertly trained operator were needed to generate a laser source beyond 1200 nm. Today, with great advances in laser technology, you can generate tremendous laser power at a long wavelength from a turnkey ultrafast pulsed laser, such as the Spectra-Physics InSight® X3.
Expand Your Research Capabilities Beyond Fluorescence Microscopy
Equipped with leading-edge microscopy and laser technologies, our FVMPE-RS multiphoton imaging system gives you the opportunity to have multiphoton, SHG, and THG microscopy all in one multimodal imaging platform. With these powerful capabilities, you can expand your research capabilities beyond fluorescence microscopy.
Related Content
Video: FVMPE-RS Multiphoton Microscope Demo
Webinar: Strategies for Deep Imaging with Multiphoton Microscopy