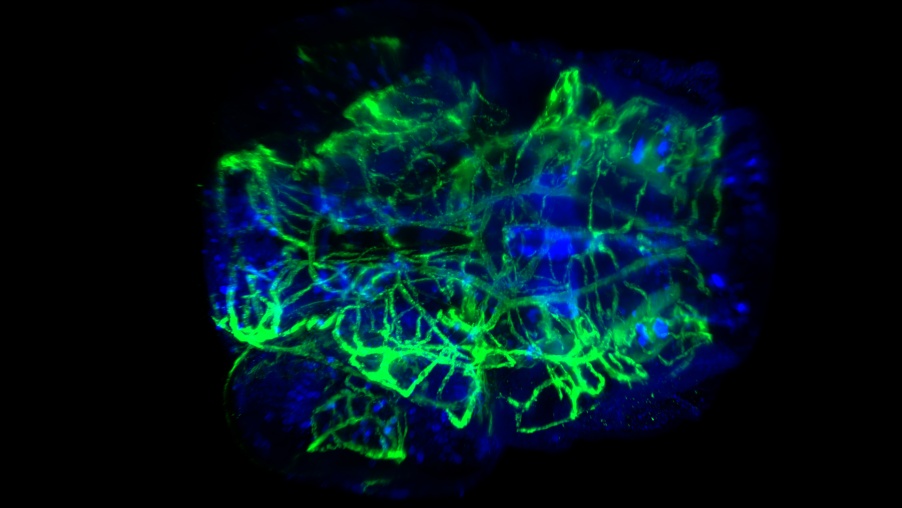
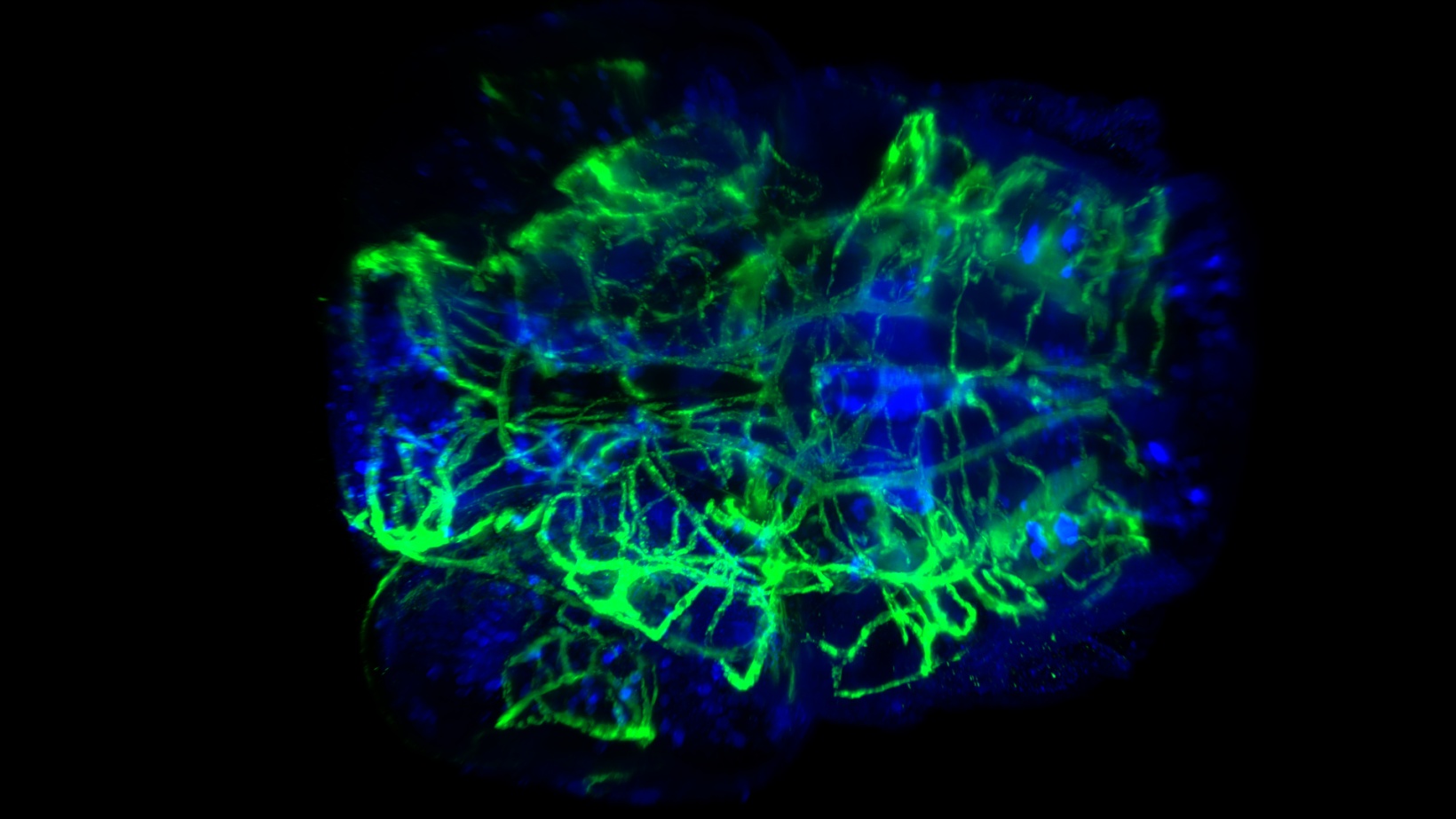
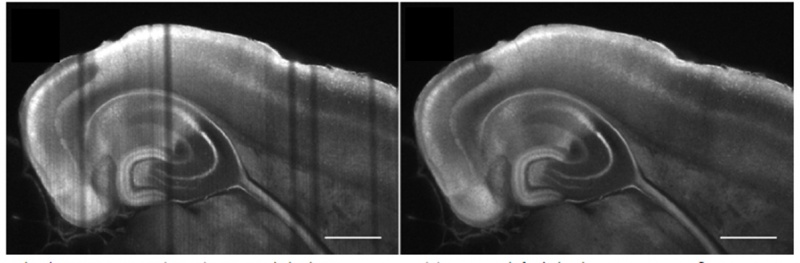
透明化したゼブラフィッシュ幼虫の頭部血管と神経の染色。20倍率のNA0.5で、CUBIC-2を用いて撮影。画像提供:フランス、Tefor FacilityのP. Affaticati氏と A. Jenett氏
私たちは、絶え間なく変化する立体的世界に住んでいます。しかし顕微鏡法分野では、3Dで、かつ高速で生体プロセスを観察するために必要な速度で画像化する私たちの能力はまだ十分ではありません。S/N比や光毒性には未だ大きな限界があるものの、蛍光顕微鏡法は他の光顕微鏡技術に比べ、高い特異性とコントラストを得ることができます。
共焦点顕微鏡法などの従来の方法では、焦点面内のピンホールを使って、セクショニング効果として知られている焦点外の光を取り除きます。この手法では、S/N比が高くなりますが、それでもサンプルは繰り返し多くの焦点外の光に暴露され、褪色しやすくなります。
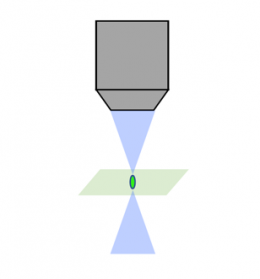 レーザー走査共焦点 | 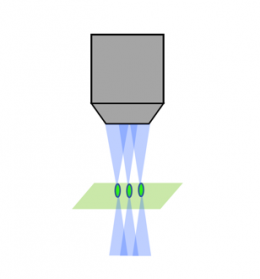 スピニングディスク共焦点 | 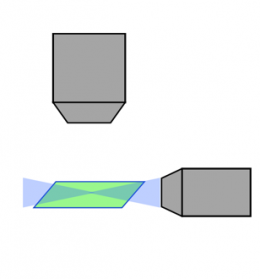 ライトシート |
そして光が見えた—ライトシート検鏡法の登場
1900年代初頭、研究者は生物学的イメージングの向上法を探っていました。その技術が1902年、Richard ZsigmondyおよびHenry Siedentopfが開発した「限外顕微鏡」と呼ばれる光学装置と共に出現しました(Heddleston & Chew, 2016; Adams et al., 2016)。この初期ライトシート顕微鏡は、照明光路と検出光路を別々にすることで従来の光学構造の型を破り、最初の直交型ライトシート顕微鏡が製作されました。
ほぼ100年後にVoieらが、光学的にモルモットの蝸牛を分割するために、直交面構造を用いた最初のライトシート蛍光顕微鏡(LSFM)画像を発表しました(Heddleston & Chew, 2016; Adams et al., 2016)。より最近では、細菌やショウジョウバエ、ゼブラフィッシュ、その他の様々な組織をイメージングするための、LSFMに変更を加えた顕微鏡が開発されています。
ライトシート蛍光顕微鏡法を大衆へ
選択的平面照明顕微鏡法(SPIM)としても知られる現代のLSFMシステムは、光学的な分割を行うために円柱レンズを使って薄いライトシートを作成してはいますが、先行製品同様、直交構造を変化させたものが採用されています。この薄いライトシートは、イメージング対象の焦点面内でサンプルの小区域のみを励起します。放出光を収集するために高速sCMOSカメラと組み合わせることで、LSFMでは、従来の共焦点技術と比較して、さらなる奥行きのサンプル、信号対雑音比の向上および光毒症を低減する速度向上が得られます。
ライトシート蛍光イメージングは現在、臓器全体または生体標本研究の重要な顕微鏡法技術として認識されています。しかし、科学界でのその開発は、現行のモデルシステムに順応する比較的限られた思考と能力により阻まれています。近年生物学分野では、研究者のニーズを満たすように「市販の」LSFMシステムがより入手しやすくなってきています。しかし全てのシステムが同じように構成されているわけではなく、様々な機能がどのように自分のニーズを満たすかを検討することが大切です。
LSFMシステムを比較する際に重要な4つの検討事項
どのライトシート蛍光システムが最適であるかを決定する際、考慮すべきいくつかの重要な点があります。
- 最初はサンプルの準備:
大きな固定されたサンプルを分割してスライドに載せる通常の従来の顕微鏡法とは異なり、ライトシート顕微鏡法のサンプルはそのままイメージングすることから、光学的に透明でなければならないため、サンプルを透過性にする必要があります。様々な組織を透明化する方法は多様で、透明化液も多く市販されています。よく使用されるいくつかの手法を確認するには、Pablo Arielの「初心者のための組織透明化ガイド(原題:A beginner’s guide to tissue clearing)」(Int J Biochem Cell Biol.2017 Mar.84:35–39)がお勧めです。 - 光学系の構造:
照明と検出光路の光学系の構造は、観察できるサンプルの性質と大きさ、さらに画質に大きな影響を与えることがあります。例えば、縞模様が入った生成物は、放射光がサンプル内部の不透明な構造で屈折したことに起因します。これを解決するため、多くの光学系構造が、あらゆる大きさの生体サンプルまたは透明化されたサンプルの画像を取得するように設計されています。このような構造の一例が、複数の照明光路を重ねてサンプルを照射し、生成物に縞模様が発生することを防ぐ多方向SPIM(mSPIM)です。
mSPIMシステムは特定のアプリケーションで最適な性能を発揮するかもしれませんが、いずれのシステムでも自由度、モジュラー対応性および実用性には制限があるため、自分の研究に最適なシステムを選ぶ際にはこれらを考慮する必要があります。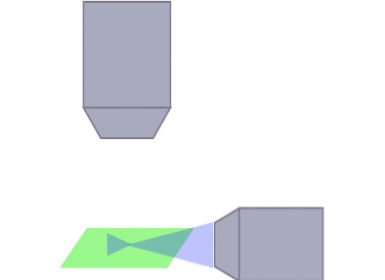
SPIM
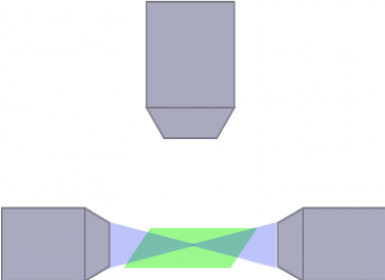
mSPIM
(Schwarz, et al.PLoS One, 2015)
- サンプルの柔軟性:
LSFMは毎年新たなシステムが市場に登場し、急速に成長している分野ですが、様々な設計が存在するため、対応できるサンプルの種類を踏まえると、非常に多様だといえます。スフェロイドなど小さめのサンプルのみのイメージングが可能なシステムもあれば、大きな組織のイメージングが可能なシステムもあります。固定標本のイメージングか、生体標本のイメージングかに限られている場合もあります。1種のサンプルのみを扱う単一ユーザーの場合はこうした特殊性が理想的なこともありますが、研究部門全体で機器を共有する、または研究を広げるための機能は低くなってしまいます。
LSFMシステムが対応する必要があると考えられる、全てのサンプル種を考慮することが大切です。最近では、サンプルの差異のギャップを埋める、自由度の高いLSFMシステムが多く設計されています。例えば、Alpha3 ライトシート蛍光顕微鏡,は、生きた生物を含む、単細胞からマウスの脳全体まで、幅広い種類の標本に対応できます。 - データ管理:最終的な、おそらく最も重要な工程は、データ管理です。1つの実験で、データが数百ギガバイト(GB)、さらには数百テラバイト(TB)に達することは珍しいことではありません。地域のITチームと協力して、自分のデータのニーズに対する計画を立てることを強く推奨します。ただし、そのような大きなデータセットを扱い、解析するには、別売の解析ソフトパッケージを購入しなければならない場合がありますので、注意が必要です。
関連コンテンツ
創造の世紀 — オリンパスのライフサイエンスイメージングシステムのストーリー
*Alpha3システムは発売されていない地域もあります。詳細については、最寄りのオリンパス代理店までお問い合わせください。