As more light sheet fluorescence microscope systems (LSFM) become commercially available, we are entering an exciting era in biological imaging. We are quickly moving away from the old standard of sectioning and mounting tissues onto slides for imaging to now capturing images of whole organisms and tissues in real time.
This post will explore all you need to know about light sheet microscopy—how it works, its key advantages, and five ways it is being used to push the boundaries of life science research.
What Is Light Sheet Microscopy?
In light sheet microscopy, the specimen is excited locally with a thin sheet of light instead of a focused spot. The emitted light is collected immediately by a detection objective that is positioned perpendicular to the illumination axis.
This configuration is commonly referred to as selective plane illumination microscopy (SPIM). Optical sectioning in this configuration is achieved at the illumination plane because only fluorophores in the field of view (FOV) are excited and there is no out-of-focus signal interference.
This enables high spatial resolution while performing volumetric imaging. As a result, a 3D stack of images can be acquired by illuminating each plane only once. This reduces the energy load on the fluorophores, resulting in reduced photobleaching and phototoxic effects.
3 Challenges of Biological Imaging with Conventional Microscopy Systems
Light sheet microscopy offers several advantages over conventional systems for various applications. Consider these common microscopy challenges:
- 3D volume rendering of data: Previous imaging technologies for large samples, such as whole mouse organs, involved physical sectioning of the organ to be imaged and then stitching data together to reconstruct the whole organ. In addition to manipulating the sample’s morphology, this causes loss of data that can be processed quantitatively.
- Slow imaging speed and reduced excitation efficiency: As mentioned in a recent blog post, confocal microscopy provides superior resolution, but at the cost of speed and excitation efficiency. Optical sectioning in this case is achieved by the presence of pinholes; however, the entire thickness of the specimen in the Z-plane is still illuminated with light multiple times, which can cause photobleaching. The sample is scanned one point at a time, so rendering the acquisition of volumetric images takes a long time. The imaging speed in confocal microscopy is improved by the presence of resonant scanners or GaAsP photomultipliers or by using a spinning disk configuration. Yet, faster scanning means fewer fluorophores are excited and fewer photons emitted, so the signal-to-noise ratio is reduced. Both confocal and spinning disk microscopy work well for fixed-tissue slices of limited thickness and time-lapse imaging of selected areas in living samples, but high-resolution imaging deep into biological tissues is inefficient.
- Expensive equipment investment: Multiphoton imaging offers high resolution and deep penetration into tissues, but this method is expensive and takes a long time to image a given tissue.
With the growing need to image live samples efficiently in 3D with low-phototoxicity, Husiken et al. 2004 built one of the first SPIM-based LSFM systems to study live processes in Medaka fish and embryogenesis in Drosophila.
Since then, light sheet systems have become a popular choice for a wide variety of applications.
5 Ways Light Sheet Microscopy Is Breaking New Ground in Life Science Research
As this imaging technique has become more mainstream in the past few years, light sheet technology has expanded its use in biological research. Here are some cool studies that biologists are working on with LSFM systems:
1. Imaging whole neural networks in a mouse brain.
Several labs are now using light sheet microscopy to image entire neuronal networks in the brains of model organisms, such as mice.
The mouse brain is cleared (made transparent through the removal of opaque particles such as lipids and proteins) and stained for neuronal markers, such as c-Fos. Using LSFM systems, the entire intact brain can be imaged without post-acquisition reconstruction, as shown in the video below:
Imaging of a whole mouse brain labelled with c-Fos at 4X magnification. Courtesy of Genaro Hernandez and David Mangelsdorf, UT Southwestern Medical Center.
This method makes it possible to quantitatively assess various parameters, such as the length of neurons, the number of branching points, and the number of dendritic spines in the neuronal network.
Consider this study: Dodt et al. (2007) used a LSFM SPIM setup with dual illumination to image whole mouse brains. They were able to visualize the shape of the hippocampus and observe the anatomical details of the whole brain. Further, they could discern neuronal somata and dendrites in the hippocampus, as well as visualize the complex network of pyramidal neurons. At 20X magnification, they also could identify dendritic spines—an important capability for neurological research:
- Dendritic spines are complex structures that are affected during learning, and spine density is known to alter in many neurological conditions such as autism, fragile X syndrome, and Alzheimer’s disease (Chidambram et al. 2019).
- Careful analyses can now be done to study dendritic spine densities and other parameters in the neuronal network of a mouse brain and compare them in disease models, thus improving our knowledge of various neurological diseases (Dodt et al. 2015).
Light sheet microscopy is also helpful for studying the distribution of neuronal activity that drives behavior, like fear. Neuronal populations are involved in the development, maintenance, and expression of fear memories in the brain (Kim et al. 2015). Currently, functional magnetic resonance imaging (fMRI) is used to determine which areas of the brain are involved in each function, but these are limited technologically as they don’t offer cellular resolution (Dodt et al. 2015).
With recent advances in LSFM, a whole mouse brain can be imaged while labelled with transcription markers for early gene activation, such as c-Fos, thus marking the functional aspects of the brain (Kim et al. 2015).
2. Visualizing vasculature in whole organs.
A crucial application of LSFM is the visualization of whole vascular networks in intact organs or tumors in model organisms such as mice. Imaging the whole vasculature structure in organs enables the quantification of parameters, such as fractional vascular volume, normalized vascular length, and mean diameter of microvessels (see the video below).
Imaging of vasculature in a mouse brain at 10X magnification.
Vascular volume and vascular length are indicators of vascular density, which is known to change in various conditions such as vascular injury, ischemia, or angiogenesis. Changes in vessel diameter are also seen in pathophysiology of several disorders, such as strokes.
To visualize the microvascular networks in whole mouse brains, Jährling et al. (2009) employed a dual illumination light-sheet setup, also known as dSPIM. The endothelial cells were stained using tomato lectin from Lycopersicon esculentum (LEA) and conjugated with FITC fluorescence dye. They used LEA because it mainly labels the endothelium of capillaries, while the staining of larger branches is less intense. Imaging lectin-labelled mouse brains resulted in visualization of even the finest branches of the microvascular network. They also obtained images of the spinal cord and the auricles of the heart, where the microvascular architecture in the cardiac auricle formed by close loops of capillaries is visible.
This approach can be applied widely in cancer research since tumors require an extensive growth of blood vessels to survive (Dodt et al. 2015). In neurobiology, studies of vasculature in the brain can reveal defects in the blood-brain barrier, which is affected by several neurological conditions, such as Alzheimer’s disease, Parkinson’s disease, amyotrophic lateral sclerosis (ALS), and multiple sclerosis, among others (Eredener Dalkara 2019, Boero 1985).
3. Calcium imaging in live brain slices.
Calcium ions generate intracellular signals that are involved in a wide variety of functions in many cell types in organisms, including regulation of gene transcription, the cell cycle, heart muscle contraction, and neurotransmitter release (Grienberger Konnerth 2015).
Imaging calcium signaling has been challenging using conventional microscopy methods because of the fast speeds at which these dynamics occur. Confocal and multiphoton microscopy provide the spatial resolution to study these changes but impose limitations on imaging speeds due to spatial scanning. In addition, these methods may introduce artifacts in reconstituted images due to the desynchronized pixel sampling (Pham et al. 2020).
Researchers today are turning to light sheet microscopy to overcome these challenges.
For example, Pham et al. (2020) used the Alpha3 light sheet microscope* to study astrocyte calcium dynamics in live mouse brain slices (see Figure 1 below). They used an agarose block to hold the live coronal brain slices in the imaging chamber, and observed distinct Ca2+ kinetics between cortical and hypothalamic astrocytes in resting condition, and on response to GPCR activation, which suggests that distinct and heterogeneous subpopulations exist with different functional and molecular properties.
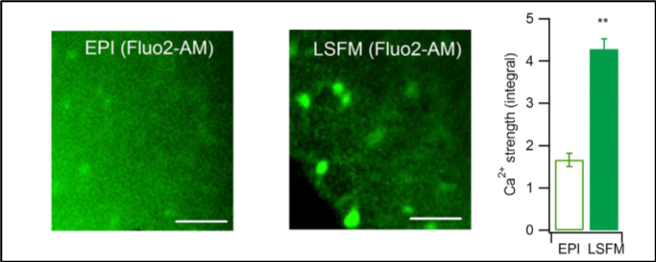
Figure 1. Imaging of calcium signal in astrocytes in acute mouse brain slices using epifluorescence (EPI) and Alpha3 light sheet (LSFM) microscopy (Pham et al. 2020).
This also suggests the importance of astrocytes in brain development and function, as astrocytes were initially thought to only play supportive roles for the nervous tissue (Grienberger Konnerth 2015). This is an example of how LSFM can be used for high-speed, volumetric functional imaging of indicators of cellular activity in tissues of considerable thickness (Hillman et al. 2019).
4. Visualization of optic region development in live teleost fish.
Teleost models are used to study morphogenesis as they are amenable to live imaging due to their external transparency.
For instance, Devos et al. (2019) used the Alpha3 light sheet microscope to compare the differences in eye morphogenesis, morphology, and patterning in the development between the two Astyanax morphs: the surface dwellers and cave dwellers. The study aimed to examine the defects in these cave fish and the mechanisms of eye morphogenesis in general.
Light sheet microscopy offered several advantages to study this development process. The wide dynamic range of the CMOS camera used in the Alpha3 light sheet system enabled detection of both strong fluorescent labelling in the telencephalon and the fainter labelling of the eye.
Besides, orthogonal dual illumination resulted in high spatial resolution without considerable photodamage as the lasers could be maintained at lower intensities. During the study, fragile live embryos were imaged for over 20 hours and developed a normal head shape at 48–60 hours post fertilization (hpf) (see Figure 2 below).
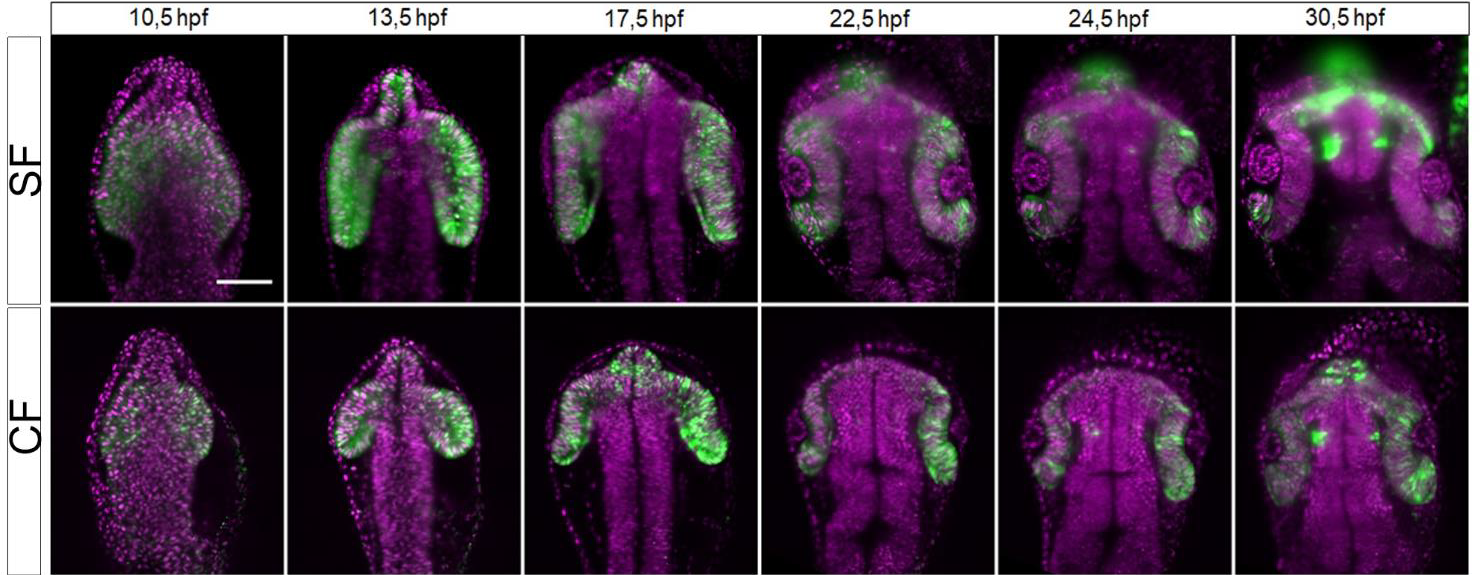
Figure 2. Live imaging of surface fish (SF) and cave fish (CF) to study eye morphogenesis. (Devos et al. 2019)
This research shows how light sheet microscopy can support the needs of biologists looking to study these developmental processes over long periods of time with minimal phototoxic effects (Wan et al. 2019).
5. High-speed imaging of whole organs in live zebrafish.
As we observed above, LSFM provides significant temporal resolution over spinning disc or confocal microscopy. Using this advantage, Lee et al. (2006) imaged embryonic hearts in live zebrafish. The hearts are relatively large, about 250 μm, and have a fast heart rate of 2–4 Hz, which makes it hard to capture it using more traditional imaging techniques.
Previous studies could achieve these speeds but only in one single plane. Consequently, getting a complete and accurate functional analysis of the heart has been a challenge until LSFM (Yalcin et al. 2017).
To accurately track myocardial wall deformations and red blood cell (RBC) movements with high resolution in the beating heart, the RBCs and myocardium were fluorescently stained. This allowed assessment of the time frame of trabeculation in a zebrafish model and established the mechanism for how shear stress activation promotes trabeculation.
The development of trabeculation is thought to be conserved between zebrafish, chicks, mice, and human embryos, so these studies can have a far-reaching impact.
*The Alpha3 light sheet microscope is available in limited countries. Contact your Olympus representative for details.
Related Content
At the Speed of Life—Light Sheet Microscopy Advances for Biological Imaging
Brochure: Alpha3 Light Sheet Microscope
A Century of Creation—The Story of Our Life Science Imaging Systems


