硅油物镜在小鼠胚胎发育过程中长时间3D活细胞成像中的应用
小鼠胚胎发育过程中的长时间3D活细胞成像
显微镜技术的进步让胚胎发育过程中的许多现象得以揭示,并成为发育生物学领域的主要研究重点。特别是,随着共聚焦显微镜的广泛使用,研究人员能够获得胚胎发育过程受精卵和单个细胞中蛋白质、DNA和其他分子的清晰三维荧光图像。
随着该领域研究的飞速发展,共聚焦显微镜已被用于长时间3D活细胞成像,以捕获随时间的动态变化以及三维荧光成像。奥林巴斯研发了可实现高对比度[PG1]长时间3D活细胞成像的硅油物镜。
本应用指南介绍了使用硅油物镜在约4天时间里将小鼠胚胎从受精卵到囊胚期的体外发育过程进行高对比度3D活细胞成像的示例。
本应用指南基于近畿大学生物科学技术学院遗传工程学系副教授Kazuo Yamagata博士及其合作者的一项研究,该研究于2014年6月发表在《干细胞报告》上。
硅油物镜在植入前发育过程MethylRO小鼠早期胚胎长时间3D活细胞成像的使用
1)获得MethylRO小鼠,将活细胞表观遗传变化进行可视化
研究人员通过将红色荧光蛋白与用于识别甲基化DNA的甲基CpG结合域蛋白1(MBD1)的甲基CpG结合域(MBD)融合获得荧光探针。然后,他们通过靶向以普遍存在基因表达而知名的ROSA26基因座而插入该基因,并获得MethylRO小鼠,这是一种在整个体内表达该探针的基因修饰小鼠品系。

图1.用于甲基化DNA可视化的新生MethylRO小鼠(黄色箭头)。
在激发光照射时,整个身体会通过滤色片(右面板)发出红色光。
2)使用60X硅油物镜对早期胚胎植入前发育过程进行长时间3D活细胞成像
Yamagata博士和他的合作者使用共聚焦显微镜在胚胎植入前大约4天的早期发育过程中对MethylRO小鼠的细胞(图1)进行延时3D活细胞成像。
研究人员将奥林巴斯硅油物镜用于活细胞观察。硅油的折射率(ne≈1.40)接近于生物组织的折射率(ne≈1.38)。油浸或水浸物镜由于与生物样品折射率不匹配造成的球差会因使用硅油物镜而降低,从而让研究人员能够在更深深度实现深层高对比度荧光成像。此外,与水浸或油浸相比,在温暖(37°C)环境使用4天以上时,硅油不会变干或变成固体,从而可以支持长时间、稳定的高分辨率3D活细胞成像。
Yamagata博士的团队以前使用油浸和水浸物镜对活细胞成像进行深度观察通过更改为硅油物镜,研究人员能够在从单细胞受精卵到囊胚期的大约4天内,观察细胞核内从表面到内部的标记荧光的甲基化DNA(mCherry-MBD-NLS)信号。
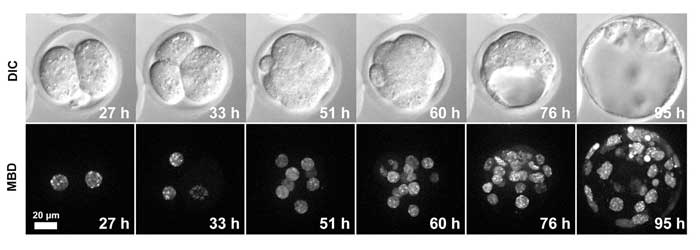
图2.植入前MethylRO胚胎发育过程中的活细胞成像。
在大约4天内观察到细胞核中甲基化DNA(mCherry-MBD-NLS)的变化。
影片1.细胞核(红色:mCherry-MBD-NLS)和细胞(绿色:CAG-EGFP)中甲基化DNA在82小时延时成像中的变化。
| |
影片2.胚泡核内甲基化DNA(mCherry-MBD-NLS)的三维图像 |
上面的图2和影片1显示,长期成像过程中的荧光漂白可以忽略不计,从而让核内甲基化DNA能够清晰成像。在影片2中,使用具有高数值孔径(1.3)和长工作距离(0.3 mm)的物镜成功获得整个100 mm胚泡的三维图像
结论:对于长时间深层、高对比度的3D活细胞成像,硅油物镜必不可少。
奥林巴斯的30/40/60 / 100X硅油物镜系列兼具高数值孔径(NA)和长工作距离。由于硅油的折射率(ne≈1.40)接近于活组织的折射率(ne≈1.38),因此在观察厚组织时,折射率不匹配导致的球差减小,从而能够实现高分辨率成像。此外,硅油不会变干,因此实验期间无需额外添加浸没液。硅油物镜可与IX电动倒置显微镜系列的Z-漂移补偿系统IX-ZDC兼容使用。利用该系统,研究人员可获得长时间保持聚焦状态且不受温度变化影响的图像。此外,硅油的折射率(ne≈1.40)和奥林巴斯提供的光学透明化试剂SCALEVIEW-A2的折射率(ne≈1.38)相当,因此使用该试剂时,折射率不匹配得到很大程度降低。当使用光学透明化样品时,具有高数值孔径的硅油物镜可提供最佳性能。
成像条件
成像系统:研究型倒置显微镜IX系列
物镜:硅油物镜UPLSAPO60XS
共聚焦扫描振镜:CSU-X1(横河电机公司)
EMCCD相机:iXON3 DU897E-CS0(安道尔科技公司)
本应用指南在近畿大学生物科学与技术学院基因工程学系副教授Kazuo Yamagata博士帮助下编写而成,
有关本应用指南所述研究的更多详细信息,请参阅以下文章:
Ueda, Jun, Kazumitsu Maehara, Daisuke Mashiko, Takako Ichinose, Tatsuma Yao, Mayuko Hori, Yuko Sato, Hiroshi Kimura, Yasuyuki Ohkawa, 和Kazuo Yamagata。“DNA甲基化报告基因小鼠MethylRO揭示了分化过程中的异染色质动力学。”干细胞报告2,第 6期(2014):910-924.
适于这类应用的产品
Maximum Compare Limit of 5 Items
Please adjust your selection to be no more than 5 items to compare at once
对不起,此内容在您的国家不适用。