Ask the Experts
Willkommen auf der Seite von Ask the Experts, dem Portal für Fragen zu Mikroskopie im Bereich der Biowissenschaften. Nehmen Sie an Live-Webinaren teil, um aktuelle Themen zu diskutieren, oder kontaktieren Sie unsere Experten bei Fragen und Herausforderungen jederzeit. Haben Sie ein Webinar verpasst? Kein Problem, Sie können es jederzeit wieder anhören. Haben Sie ein Thema, das Sie diskutieren möchten? Nutzen Sie unser Feedback-Formular oder wenden Sie sich direkt an unsere Experten.
Live-Webinare | Online-Webinare | Experten
.jpg?rev=3E0D)
Mikroskop-Objektive: Wo die Magie beginnt
Experten
In diesem Webinar mit Lauren Alvarenga und Klaus Willeke geht es um die Bedeutung einer guten Optik in einem komplexen System wie einem Mikroskop. Die beiden Referenten werden verschiedene Aspekte diskutieren, die für die endgültige Bildqualität wichtig sind.
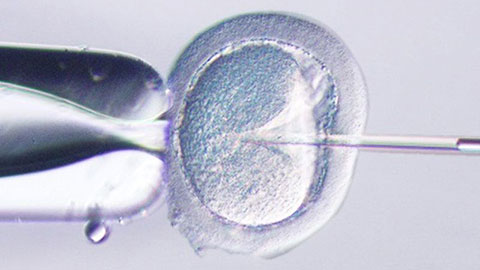
ICSI—Past, Present & Future
Experten
In this webinar, Dr. Pai will be sharing on what’s new in the management of male infertility with special focus on Intra Cytoplasmic Sperm Injection (ICSI). He will be describing the latest diagnostic modalities in male infertility especially sperm DNA fragmentation, genetic testing, cryotozoospermia and microsurgical TESA in cases of non-instructive azoospermia.
.jpg?rev=1940)
Digitale Bildverarbeitung Teil 2: Erweiterte Bildverarbeitungsfilter
Experten
In unserem ersten Webinar zu diesem Thema haben wir gelernt, dass mit einem Lichtmikroskop aufgenommene Bilder nie ein perfektes Abbild unserer Proben sind. Es gibt immer Fehlerquellen. Da wir diese Fehler nur minimieren und nicht eliminieren können, müssen die Rohdaten eines Bildgebungsexperiments vor der endgültigen Bildanalyse oft digital verarbeitet werden.
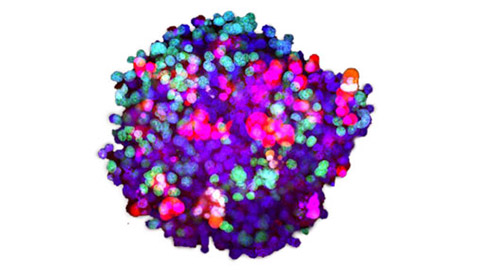
Dreidimensionale hochleistungsfähige Bildanalyse von Organoiden und Sphäroiden aus Patientenzellen
Experten
Organoide und Sphäroide können die Bedingungen in vivo besser nachbilden, und durch Bildgebungsanalysen lassen sich zellspezifische Reaktionen mit hoher räumlicher Auflösung verfolgen. Daher haben wir unter Verwendung von Krebsorganoiden und -sphäroiden aus Patientenzellen dreidimensionale Analyse- und Wirkstoffbewertungsmethoden für die Bildgebung entwickelt.
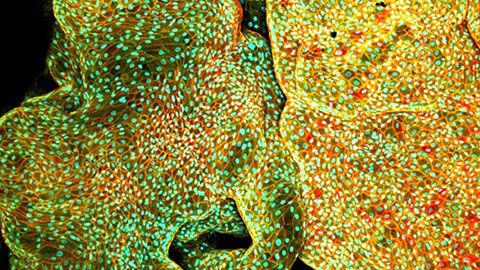
Depth Matters: Transforming Biology for More Realistic and Meaningful Pursuits
Experten
Improvements to in vitro three-dimensional (3D) models are making them increasingly better at mimicking in vivo-like cellular behavior. Every tissue presents a distinct microenvironment with a unique blend of biochemical and biophysical components that dictate cellular behavior. Recreation of critical features of tissues that nurtures recapitulation of in vivo-like cellular behavior is the essence of an effective 3D model. In this webinar, through specific examples of 3D models for tissue development and cancer, we will revisit the fundamental principles of designing 3D models that can effectively recapitulate critical features of the tissues in vitro and applications of such models in mechanistic studies and drug testing. Our work also highlights the importance of 3D imaging systems, such as laser scanning confocal microscopes, which are necessary for such work.
Olympus Organoid Conference 2021
During the Olympus Organoid Conference, cell biologists, microscopists, and image analysis experts shared their insights and answered questions on the latest developments in organoid technologies.
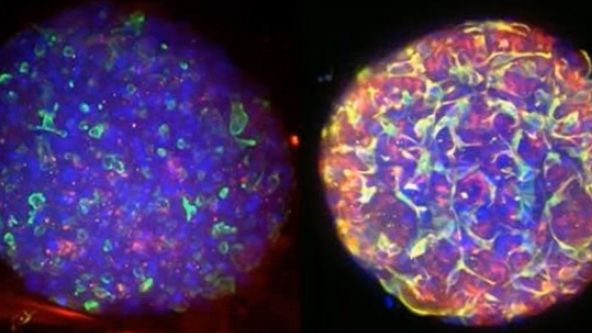
Culture and Quantitative 3D Imaging of Organoids: Challenges and Solutions
Experten
Turning organoids into impactful translational models includes being able to culture them and assess those that develop robustly with physiologically relevant architecture. However, quantitative comparisons and statistical analysis at high content, which are mandatory to describe the complexity of such multicellular 3D objects are not possible owing to the lack of high-throughput 3D imaging methods. We have thus engineered a versatile High Content Screening (HCS) device to streamline all the steps of organoid culture to exploit its potential in morphogenesis understanding. Our approach comprises a new generation of versatile scaffolding cell culture multiwell chips with embedded optical components (= lighting JeWells) that enables rapid 3D imaging.
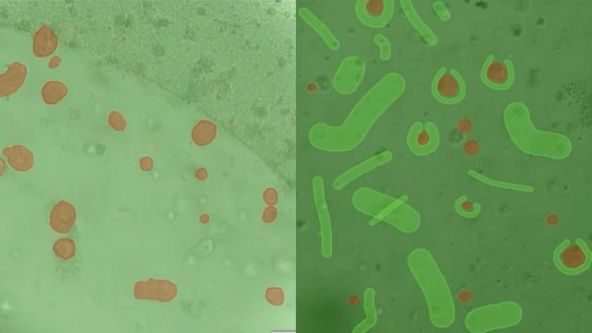
Converting from 2D to 3D: Bio-Techne Solutions for Your 3D Culture
Experten
Organoid and three-dimensional (3D) cell culture are emerging as pivotal systems for understanding human organ development, modeling disease, screening for drug efficacy or toxicity, and investigating personalized medicine. Usually they are derived from primary tissue, embryonic stem cells (ESCs), or induced pluripotent stem cells (iPSCs), which are capable of self-renewal and differentiation.
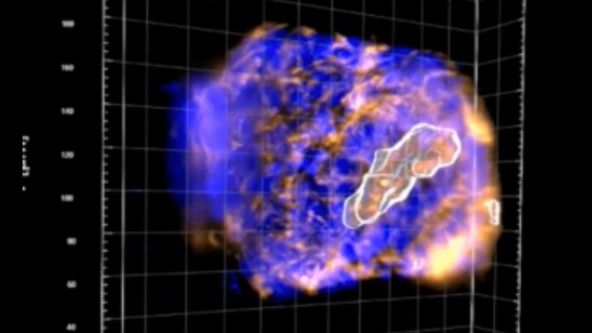
Utilizing Tumoroids to Explore Anti-Tumor Immunity in Rectal Cancer
Experten
Globally colorectal cancer is a significant public health burden. It is the third most commonly diagnosed cancer and fourth leading cause of cancer related deaths in the world. A subset of patients diagnosed with rectal cancer require neoadjuvant chemoradiotherapy (NACRT) prior to surgery. However, there is a spectrum of response to this therapy with only 10-20% of patients achieving a complete pathological response. In addition, 20-40% of patients will demonstrate no response to this treatment. There is currently no method that predicts how a patient will respond to NACRT accurately. In order to investigate the mechanisms underpinning how patients respond to therapy, patient derived tumouroids have been utilised. These personalised in vitro three-dimensional tumour models recapitulate the in vivo tumour of origin genotypically. The Ramsay laboratory (Peter MacCallum, Melbourne) has successfully co-cultured patient-matched rectal cancer tumouroids with tumour infiltrating lymphocytes (TILS) in a novel in vitro assay, preliminary data generated suggests this assay has the ability to predict the response of a patient to treatment with NACRT prior to instigation of neo-adjuvant therapy. This assay provides a pre-clinical platform that encapsulates the hosts immune response toward their tumour. However, manual analysis of the data generated from this assay is time consuming and limits the clinical utility of this platform. Machine-based learning to develop artificial neural networks capable of analysing data produced from the killing assay has been developed to automate analysis. Automated analysis utilising artificial neural networks is a feasible approach to expedite the processing of data generated from the cytotoxic killing assays and will improve the clinical utility of this platform to direct personalised patient therapy.
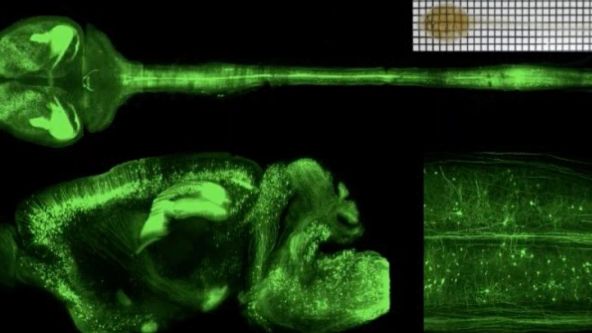
Tissue Optical Clearing Imaging: From In Vitro to In Vivo
Experten
Biomedical optical Imaging, as a powerful tool has been applied for observing biomedical tissue structural and functional information with high resolution and contrast unattainable by any other method. However, the high scattering of turbid biological tissues limits the penetration of light, leading to strongly decreased imaging resolution and contrast as light propagates deeper into the tissue. Fortunately, novel tissue optical clearing technique provide a way for reducing the scattering of tissue and improving the optical imaging quality. This presentation will introduce our progress from in vitro and in vivo of tissue optical clearing imaging, including developing in vitro optical clearing methods, such as FDISCO and MACS. Meanwhile, we will also demonstrate in vivo skull/skin optical clearing window for imaging structural and functional of cutaneous / cortical vascular and cells, also manipulating cortical vasculature.
3D Microscopy: Understanding the Give and Take on Instrument Performance to Enable Informed Decisions
Experten
Biologists have a significant toolbox at their disposal when it comes to microscopically imaging 3D samples, such as organoids. From widefield microscopy to confocal, superresolution, multiphoton and lightsheet, each have their own set of pros and cons that must be carefully considered before making an informed choice on the most suitable to address your biological question. Often a correlative approach is required, applying several techniques to address the question from different perspectives. It is also crucial to consider the method of sample preparation and optimise each of the potential steps which can include fixation, permeabilisation, labelling and mounting. Further, the images generated by all techniques can be enhanced with post-processing techniques, such as deconvolution, which can enable or help to improve subsequent image analysis and interpretation.
Advances in 3D Optical Imaging Technologies: An Overview
Experten
With rapid development in fluorescent proteins, synthetic fluorochromes, and digital imaging, advanced 3D imaging technologies are now available to investigators to provide critical insights into the fundamental nature of cellular and tissue functions. 3D and 4D imaging systems have become very common tools among biologists. However, there are several technical challenges and limitations in performing successful 3D and 4D imaging. Olympus has developed a wide range of 3D imaging microscopes to overcome these challenges and to satisfy the requirements of researchers across different disciplines.












