Not Available in Your Country
Sorry, this page is not
available in your country.
Overview
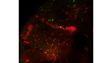
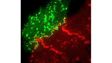
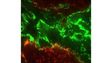
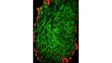
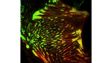
 | Excellent Multicolor TIRF ImagingDesigned for membrane dynamics, single molecule detection, and colocalization experiments, the IXplore TIRF microscope system offers simultaneous multicolor TIRF imaging for up to 4 colors with high stability. |
|---|
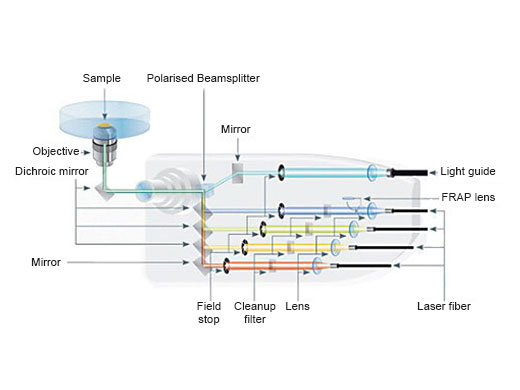
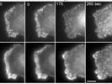
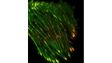
| Simultaneous Multicolor TIRFThe Olympus cellTIRF system provides true simultaneous acquisition of up to four wavelengths.
|
|---|
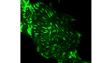
Imaging StabilityThe frame architecture and focus drive design of the IXplore system offer enhanced rigidity that reduces the impact of vibration and temperature. It maintains the desired Z-axis position to facilitate reliable time-lapse imaging. When combined with the TruFocus system, the IXplore TIRF microscope system can capture high-precision, time-lapse images that are aligned and in focus. | Related Videos |
| TIRF ObjectivesTotal internal reflection fluorescence (TIRF) is facilitated by a wide range of objectives featuring a high signal-to-noise ratio and a correction collar to adjust for cover glass thickness and temperature. Our corrected plan apochromat objectives with an NA of 1.5 help you acquire uniform high-quality images with a large field of view. Take advantage of Olympus' remarkable TIRF objective with the world’s highest NA, 1.7*. *As of November, 2018. According to Olympus research. |
|---|
Precise and Intuitive Photomanipulation (Optional Peripherals)Olympus’ cellFRAP photomanipulation device and real-time controller (U-RTCE) enable accurate control (200 µs dead time), diffraction-limited stimulation with a flexible region of interest, and precise reproduction of experimental conditions. |
|
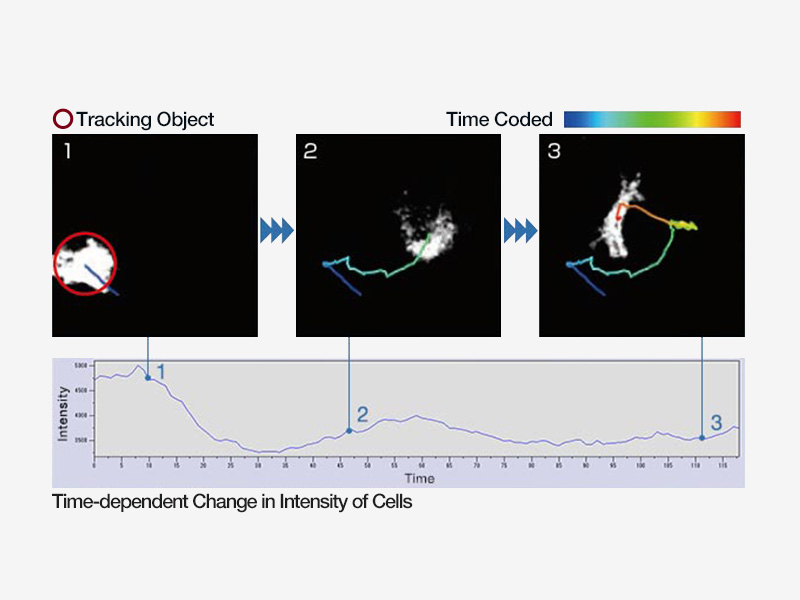 | Advanced AnalysisAdvanced functionality, such as object tracking, Count and Measure, Kymograph and real time 2D deconvolution to enhance object identification. |
|---|
Fast, Microsecond-Accurate DevicesThe fast filter wheel, shutter, LED light source control, and real-time controllers (U-RTC) enable less photobleaching and phototoxicity, resulting in healthier cells and more robust data. | Related Videos |
ReferencesY. Yang, et al. Spectraplakin induces positive feedback between fusogens and the actin cytoskeleton to promote cell-cell fusion. Developmental Cell (April 10, 2017). A. R. van Vliet, et al. The ER stress sensor PERK coordinates ER-plasma membrane contact site formation through interaction with filamin-A and F-actin remodeling. Molecular Cell (Feburuary 23, 2017). F. Hertel, et al. RefSOFI for mapping nanoscale organization of protein-protein interactions in living cells. Cell Reports (December 31, 2015). C. Cauvin, et al. Rab35 GTPase triggers switch-like recruitment of the lowe syndrome lipid phosphatase OCRL on newborn endosomes. Current Biology (December 24, 2015). W.-K. Ji, et al. Actin filaments target the oligomeric maturation of the dynamin GTPase Drp1 to mitochondrial fission sites. eLIFE (November 26, 2015). A. Juanes-Garcia, et al. A regulatory motif in nonmuscle myosin II-B regulates its role in migratory front–back polarity. The Journal of Cell Biology (April 13, 2015). D. Borrenberghs, et al. HIV virions as nanoscopic test tubes for probing oligomerization of the integrase enzyme. ACS Nano (March 21, 2014). S. Yamaoka, et al. Identification and dynamics of arabidopsis adaptor protein-2 complex and its involvement in floral organ development. The Plant Cell (August 23, 2013) |
Need assistance? |
Specifications
| Microscope Frame | IX83P2ZF | |
|---|---|---|
| Observation Method > Total Internal Reflection Fluorescence | ✓ | |
| Observation Method > Fluorescence (Blue/Green Excitation) | ✓ | |
| Observation Method > Fluorescence (Ultraviolet Excitation) | ✓ | |
| Observation Method > Differential Interference Contrast (DIC) | ✓ | |
| Observation Method > Phase Contrast | ✓ | |
| Observation Method > Brightfield | ✓ | |
| Revolving Nosepiece > Motorized (6 position) | ✓ | |
| Focus > Motorized |
| |
| Focus > Z Drift Compensator | ✓ | |
| Observation Tubes > Widefield (FN 22) > Tilting Binocular | ✓ | |
| Illuminator > Transmitted Köhler Illuminator > LED Lamp | ✓ | |
| Illuminator > Transmitted Köhler Illuminator > 100 W Halogen Lamp | ✓ | |
| Illuminator > Fluorescence Illuminator > 100 W Mercury Lamp | ✓ | |
| Illuminator > Fluorescence Illuminator > Light Guide Illumination | ✓ | |
| Fluorescence Mirror Turret > Motorized (8 position) | ✓ | |
| Stage > Motorized | Contact your local sales representative to hear about motorized stage options | |
| Condenser > Motorized > Universal Condenser | W.D. 27 mm, NA 0.55, motorized aperture and polarizer | |
| Condenser > Manual > Universal Condenser | NA 0.55/ W.D. 27 mm | |
| Condenser > Manual > Ultra-Long Working Distance Condenser | NA 0.3/ W.D. 73.3 mm | |
| Confocal Scanner | - | |
| Super Resolution Processing | - | |
| Accessories | Remote correction collar controller (IX3-RCC) | |
| Dimensions (W × D × H) | 323 (W) x 475 (D) x 706 (H) mm (IX83 microscope frame) | |
| Weight | Approx. 47kg (IX83P2ZF) |